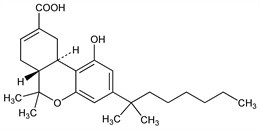
198. Ajulemic Acid
Nomenclature
Description and references
Cannabinoid receptor agonist; synthetic, nonpsychoactive derivative of Δ9-tetrahydrocannabinol, q.v. Prepn: S. H. Burstein et al., J. Med. Chem. 35, 3135 (1992); S. H. Burstein, R. Mechoulam, WO 9401429; eidem, US 5338753 (both 1994). GC-MS determn in plasma: C. Batista et al., J. Chromatogr. B 820, 77 (2005). Binding to CB-receptors: M.-H. Rhee et al., J. Med. Chem. 40, 3228 (1997); to PPARγ receptor: J. Liu et al., Mol. Pharmacol. 63, 983 (2003). Clinical evaluation in chronic neuropathic pain: M. Karst et al., J. Am. Med. Assoc. 290, 1757 (2003). Reviews of development and pharmacology: S. Burstein et al., Life Sci. 75, 1513-1522 (2004); idem, AAPS J. 7, E143-E148 (2005); J. L. Wiley, IDrugs 8, 1002-1011 (2005).
Properties
Crystals from acetonitrile, mp 112-114° (sintering). [α]D 275° (c = 3.8 in chloroform). Sol in most organic solvents. Practically insol in hexane.Derivative
Acetate.Properties
Crystals from pentane, mp 120-122°. [α]D 265° (c = 9.0 in chloroform).Therapeutic Category
Anti-inflammatory.
Keywords
Anti-inflammatory (Nonsteroidal)