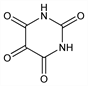
Nomenclature
CAS number: 50-71-5
2,4,5,6(1
H,3
H)-pyrimidinetetrone; 2,4,5,6-tetraoxohexahydropyrimidine; mesoxalylurea; mesoxalylcarbamide.
C
4H
2N
2O
4; mol wt 142.07.
C 33.82%, H 1.42%, N 19.72%, O 45.05%.
Description and references
Found by Liebig in mucus excreted during dysentery.
Prepn by direct oxidation of uric acid: G. Brugnatelli, Ann. Chim. Phys. [2] 8, 201 (1818); J. Liebig, F. Whler, Ann. 26, 241 (1838); H. Biltz, M. Hehn, ibid. 413, 60 (1917). Prepd from alloxantin: J. Liebig, ibid. 147, 366 (1868); W. W. Hartman, O. E. Sheppard, Org. Synth. coll. vol. III, 37
(1955). See also A. V. Holmgren, W. W. Wenner, ibid. coll. vol. IV, 23 (1963). Produces diabetes in animals
by selective necrosis of pancreatic islet β-cells: J. S. Dunn, N.
G. B. McLetchie, Lancet 2, 384 (1943); W. B. Kennedy, F. D. W. Lukens, Proc. Soc. Exp. Biol. Med. 57, 143 (1944).
Mechanism of action study: L. Boquist, Acta
Pathol. Microbiol. Scand. Sect. A 88, 201 (1980).
In vitro antineoplastic activity: P. Grobon, C. R. Seances Acad. Sci. Ser. D 280, 2413 (1975). Antibacterial and antifungal activity: J. D. Douros,
A. F. Kerst, JP Kokai 72 4900; eidem, US 3728454 (1972, 1973 both to Gates
Rubber Co.). Toxicological study in mice: B. A.
Waisbren, Proc. Soc. Exp. Biol. Med. 67, 154 (1948).
Properties
Anhydrous, orthorhombic crystals from anhydr acetone
or glacial acetic acid or by sublimation in vacuo. Turns pink at
230° and dec at 256°. Acid to litmus. pK (25°) 6.63. Absorption spectrum: Hartley, J. Chem. Soc. 87, 1802, 1808 (1905).
Freely sol in water. Hot aqueous solns are yellow and become colorless
on cooling. Aqueous solns, after being in contact with the human
skin for some time, give it a red color and a disagreeable odor.
Sol in acetone, alcohol, methanol, glacial acetic acid; slightly sol
in chloroform, petr ether, toluene, ethyl acetate and acetic anhydride.
Insol in ether.Derivative
Tetrahydrate.
Properties
Large triclinic prisms or oblique monoclinic rhombs
from water.Derivative
Monohydrate.
Properties
By heating the tetrahydrate at 100° or by exposing
it to the air. Forms triclinic pinacoidal crystals.Derivative
Compound with urea.
CH
4N
2O.C
4H
2N
2O
4.H
2O; mol wt 220.14.
C 27.28%, H 3.66%, N 25.45%, O 43.61%.
Properties
Minute, yellow needles; red at 170°, decomp at
185-186°.Use
In production of diabetes in experimental animals;
in nutrition experiments; in organic syntheses.
