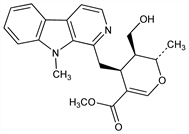
315. Alstonidine
Nomenclature
CAS number: 25394-75-6
(2S,3S,4S)-3,4-Dihydro-3-(hydroxymethyl)-2-methyl-4-[(9-methyl-9H-pyrido[3,4-b]indol-1-yl)methyl]-2H-pyran-5-carboxylic
acid methyl ester; 3β-(hydroxymethyl)-2α-methyl-4β-[(9-methyl-9H-pyrido[3,4-b]indol-1-yl)methyl]-2H-pyran-5-carboxylic acid
methyl ester. C22H24N2O4; mol wt 380.44.
C 69.46%, H 6.36%, N 7.36%, O 16.82%.
Description and references
From the bark or root of Alstonia constricta F. Muell., Apocynaceae: Hesse, Ann. 205, 360 (1880); Svoboda, J. Am. Pharm. Assoc. 46, 508 (1957). Structure: Boaz et al., ibid. 510. Stereochemistry: Crow et al., Aust. J. Chem. 23, 2489 (1970).
Properties
Fine crystals from ether, mp 188-190°. Practically insol in water. Sol in alcohol, chloroform, ether, acetone, 5% HCl. uv max (methanol): 238, 291, 360 nm (log ε 4.66, 4.25, 3.74). The acid solutions are fluorescent.Derivative
Acetyl deriv trihydrate.C24H26N2O5.3H2O; mol wt 476.52.
C 60.49%, H 6.77%, N 5.88%, O 26.86%.