374. Amantadine
Nomenclature
Description and references
NMDA-receptor antagonist. Antiviral agent active vs influenza A and other RNA viruses; interacts with the M2 viral membrane matrix protein, blocking the release of viral RNA into the cytoplasm of infected cells. Prepn: H. Stetter et al., Ber. 93, 226 (1960) DOI; W. Haaf, ibid. 97, 3234 (1964) DOI; P. Kovacic, P. D. Roskos, Tetrahedron Lett. 1968, 5833. LC-MS/MS determn in serum: T. Arndt et al., Clin. Chim. Acta 359, 125 (2005) DOI PubMed. Antiviral activity: W. L. Davies et al., Science 144, 862 (1964) DOI PubMed. Pharmacology and toxicology: V. G. Vernier et al., Toxicol. Appl. Pharmacol. 15, 642 (1969) DOI PubMed. Comprehensive description: J. Kirschbaum, Anal. Profiles Drug Subs. 12, 1-36 (1983). Review of use vs influenza A: R. L. Tominack, F. G. Hayden, Infect. Dis. Clin. North Am. 1, 459-478 (1987) PubMed; of pharmacokinetics: F. Y. Aoki, D. S. Sitar, Clin. Pharmacokinet. 14, 35-51 (1988) DOI PubMed. Review of NMDA receptor binding and neuroprotective properties: J. Kornhuber et al., J. Neural Transm. 43, Suppl., 91-104 (1994). Series of articles on clinical experience in Parkinson's disease: ibid. 46, Suppl., 399-421 (1995). Review of therapeutic potential in hepatitis C infection: J. K. Lim et al., J. Viral Hepat. 12, 445-455 (2005) DOI PubMed.
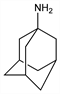
Properties
Crystals by sublimation, mp 160-190° (closed tube) (Stetter). Also reported as mp 180-192° (Haaf). pKa: 10.1. Sparingly sol in water.Derivative
Hydrochloride.Nomenclature
Properties
Crystals from abs ethanol + anhydr ether, mp >360° (dec). Freely sol in water (at least 1:20); sol in alcohol, chloroform. Practically insol in ether. LD50 orally in mice, rats: 700, 1275 mg/kg (Vernier).Derivative
Sulfate.Nomenclature
Therapeutic Category
Antiviral; antiparkinsonian.
Therapeutic Category (Veterinary)
Antiviral; adjunct for chronic pain.Keywords
Antidyskinetic; Antiparkinsonian; Antiviral; Adamantanes