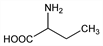
428. α-Aminobutyric Acid
Nomenclature
CAS number: 2835-81-6
2-Aminobutanoic acid; α-amino-n-butyric acid. C4H9NO2; mol wt 103.12.
C 46.59%, H 8.80%, N 13.58%, O 31.03%.
Description and references
Prepn of the dl-form from α-bromobutyric acid and ammonia: Fisher, Mounegrat, Ber. 33, 2388 (1900); from potassium cyanide, ammonium chloride and propionaldehyde: Zelinsky, Stadnikow, Ber. 41, 2062 (1908); by reduction of α-oxobutyric acid: Knoop, Oesterlin, Z. Physiol. Chem. 148, 305 (1925); from α-ethylacetoacetic ester and hydrazoic acid: Schmidt, Ber. 57, 706 (1924). The l(+)-form has been isolated from proteins: Oikawa, Chem. Zentralbl. 1926, 1, 148. Configuration: Clough, J. Chem. Soc. 113, 544, 551; Levene, Chem. Rev. 2, 203 (1926); Vogler, Helv. Chim. Acta 30, 1766 (1947).