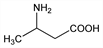
429. β-Aminobutyric Acid
Nomenclature
Description and references
Prepd from pyrotartaric acid diamide: Weidel, Roithmer, Monatsh. Chem. 17, 185 (1896); from β-chlorobutyric acid ethyl ester and alcoholic NH3: Balbiano, Ber. 13, 312 (1880); from crotonic acid and concd NH3: Engel, Bull. Soc. Chim. [2] 50, 102 (1888); Curtius, Gumlich, J. Prakt. Chem. [2] 70, 204 (1904); Stadnikow, Chem. Zentralbl. 1909, II, 1988; see Fischer, Roeder, Ber. 34, 3755 (1901) footnote; Stoermer, Robert, Ber. 55, 1038 (1922). Prepn of HCl salt from β-aminobutyronitrile: Bruylants, Bull. Soc. Chim. Belg. 32, 259 (1923); Chem. Zentralbl. 1924, I, 1668. By reducing acetoacetic ester phenylhydrazone and saponifying the resulting ester: Fischer, Groh, Ann. 383, 338 (1911). The d()-form has been obtained by hydrolysis of its ester: Fischer, Scheibler, ibid. 346.