470. p-Aminopropiophenone
Nomenclature
CAS number: 70-69-9
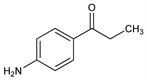
1-(4-Aminophenyl)-1-propanone; ethyl p-aminophenyl ketone; PAPP. C9H11NO; mol wt 149.19.
C 72.46%, H 7.43%, N 9.39%, O 10.72%.
Description and references
Pretreatment antidote to cyanide; activity probably through the formation of methemoglobin, which has a higher affinity for cyanide than mitochondrial cytochrome oxidase. Prepd by the action of propionyl chloride on aniline in carbon bisulfide in the presence of aluminum chloride: Kunckell, Ber. 33, 2641 (1900); Derrick, Bornemann, J. Am. Chem. Soc. 35, 1283 (1913); Hartung, Foster, J. Am. Pharm. Assoc. 35, 15 (1946). Antidotal activity: C. L. Rose et al., J. Pharmacol. Exp. Ther. 89, 109 (1947). Toxicity study: J. W. Scawin et al., Toxicol. Lett. 23, 359 (1984). Review of pharmacology of cyanide detoxification: S. I. Baskin, R. F. Fricke, Cardiovasc. Drug Rev. 10, 358-375 (1992).
Properties
Yellow needles from water, mp 140°. Sol in water, alc, DMSO. LD50 in male, female mice (mg/kg): 145, 200 i.v.; in female mice, guinea pigs, rats, male rats (mg/kg): >5000, 1020, 223.7, 475 orally (Scawin).Derivative
Hydrochloride.Nomenclature
CAS number: 6170-25-8
C9H11NO.HCl; mol wt 185.65.
C 58.23%, H 6.52%, N 7.54%, O 8.62%, Cl 19.10%.
Properties
Needles, yellowish cast, mp 198-199°. Freely sol in water. LD50 i.v. in dogs: 7.15 ±0.89 mg/kg (Rose).Derivative
Sulfate.(C9H11NO)2.H2SO4; mol wt 396.46.
C 54.53%, H 6.10%, N 7.07%, O 24.21%, S 8.09%.
Properties
Yellow plates from alcohol, dec 25°.Therapeutic Category
Antidote (cyanide).
Keywords
Antidote (Cyanide)