472. Aminopterin
Nomenclature
Description and references
Folic acid antagonist. Prepd from 2,4,5,6-tetraminopyrimidine sulfate, 2,3-dibromopropionaldehyde and p-aminobenzoylglutamic acid: D. R. Seeger et al., J. Am. Chem. Soc. 69, 2567 (1947) DOI; from 6-(bromomethyl)-2,4-diaminopteridine HBr: J. R. Piper, J. A. Montgomery, J. Heterocycl. Chem. 11, 279 (1974) DOI. Purification: T. L. Loo, J. Med. Chem. 8, 139 (1965) DOI. Inhibition of dihydrofolate reductase: J. S. Erickson et al., J. Biol. Chem. 247, 5661 (1972) PubMed. Use as rodenticide: US 2575168 (1951 to Am. Cyanamid). Clinical pharmacokinetics: A. F. Ratliff et al., J. Clin. Oncol. 16, 1458 (1998) PubMed. Clinical evaluation in children with acute lymphoblastic leukemia (ALL): P. D. Cole et al., Cancer Chemother. Pharmacol. 62, 65 (2008) DOI PubMed.
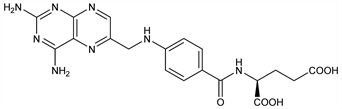
Derivative
Dihydrate.Properties
Clusters of yellow needles. uv max of the 0.75 hydrate (0.1N NaOH): 261, 282, 373 (log ε 4.41, 4.39, 3.91).Use
Rodenticide.Therapeutic Category
Antineoplastic.
Keywords
Antineoplastic; Antimetabolites; Folic Acid Analogs/Antagonists