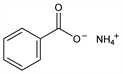
496. Ammonium Benzoate
Nomenclature
CAS number: 1863-63-4
Benzoic acid ammonium salt (1:1). C7H9NO2; mol wt 139.15.
C 60.42%, H 6.52%, N 10.07%, O 23.00%.
Description and references
It is about 99% pure. Manuf from benzoic acid and NH3: J. A. Spina, US 1704636 (1929 to Hooker Electrochem.). Analytical applications in metal cation separations: I. M. Kolthoff et al., J. Am. Chem. Soc. 56, 812 (1934); A. Jewsbury, G. H. Osborn, Anal. Chim. Acta 3, 642 (1949). Structural studies: I. A. Oxton et al., Can. J. Chem. 55, 3831 (1977).