585. Amphotericin B
Nomenclature
Description and references
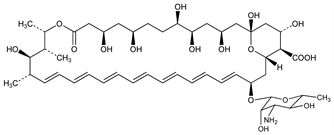
Polyene antibiotic produced by Streptomycetes nodosus M4575 obtained from soil of the Orinoco river region of Venezuela: Gold et al., Antibiot. Annu. 1955-1956, 579; Vandeputte et al., ibid. 587; Dutcher et al., ibid. 1956-1957, 866; Walters et al., J. Am. Chem. Soc. 79, 5076 (1957) DOI; Dutcher et al., US 2908611 (1959 to Olin Mathieson). Structure studies: Borowski et al., Tetrahedron Lett. 6, 473 (1965) DOI. Carbon skeleton, ring size, and partial structure: Cope et al., J. Am. Chem. Soc. 88, 4228 (1966) DOI. Complete structure: Mechlinski et al., Tetrahedron Lett. 11, 3873 (1970) DOI; Borowski et al., ibid. 3909 DOI; R. C. Pandey, K. L. Rinehart, J. Antibiot. 29, 1035 (1976) PubMed. Total synthesis: K. C. Nicolaou et al., J. Am. Chem. Soc. 109, 2821 (1987) DOI. Toxicity: G. R. Keim et al., Science 179, 584 (1973) DOI PubMed. Comprehensive description: I. M. Asher et al., Anal. Profiles Drug Subs. 6, 1-42 (1977). Review of clinical experience: H. A. Gallis et al., Rev. Infect. Dis. 12, 308-329 (1990) PubMed; of lipid formulations: J. W. Hiemenz, T. J. Walsh, Clin. Infect. Dis. 22, Suppl. 2, S133-S144 (1996) PubMed. Review: K. M. Abu-Salah, Br. J. Biomed. Sci. 53, 122-133 (1996) PubMed.
Properties
Deep yellow prisms or needles from DMF. Dec gradually above 170°. uv max (methanol): 406, 382, 363, 345 nm. [α]D24 +333° (acidic DMF); 33.6° (0.1N methanolic HCl). Sol in propylene glycol; slightly sol in methanol. Insol in anhydrous alcohol, ether, benzene, toluene. Insol in water at pH 6 to 7. Soly at pH 2 or pH 11 in water: about 0.1 mg/ml. Water soly increased by sodium desoxycholate. Soly in DMF 2 to 4 mg/ml; in DMF + HCl: 60 to 80 mg/ml; in DMSO: 30 to 40 mg/ml. Solids and solns appear stable for long periods between pH 4 and 10 when stored at moderate temps out of contact with light and air. LD50 in mice (mg/kg): 88 i.p., 4 i.v. (Keim).Derivative
Compd with cholesteryl sulfate.Nomenclature
Description and references
Reviews: L. S. S. Guo, P. K. Working, J. Liposome Res. 3, 473-490 (1993) DOI; D. A. Stevens, J. Infect. 28, Suppl.1, 45-49 (1994).
Properties
Forms uniform disc-shaped particles in aq media.Derivative
Liposomal complex.Nomenclature
AmBisome (Nexstar).Description and references
Unilamellar liposomes containing phosphatidylcholine, cholesterol, distearoylphosphatidylglycerol and amphotericin B in a molar ratio of 2: 1:0.8:0.4. Review: R. J. Hay, J. Infect. 28, Suppl. 1, 35-43.
Derivative
Amphotericin B lipid complex.Nomenclature
ABLC; Abelcet (Liposome Co.).Description and references
Complex with 2 phospholipids, l-α-dimyristoyl phosphatidylcholine and l-α-dimyristoyl phosphatidylglycerol, which are present in a 7:3 molar ratio. Review: J. Lister, Eur. J. Haematol. Suppl. 57, 18-23 (1996) PubMed.
Therapeutic Category
Antifungal.
Therapeutic Category (Veterinary)
Antifungal.Keywords
Antifungal (Antibiotics); Polyenes