616. Amylpenicillin
Nomenclature
CAS number: 4493-18-9
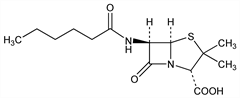
(2S,5R,6R)-3,3-Dimethyl-7-oxo-6-[(1-oxohexyl)amino]-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic
acid; dihydropenicillin F; hexanoylpenicillin; penicillin DF. C14H22N2O4S; mol wt 314.40.
C 53.48%, H 7.05%, N 8.91%, O 20.36%, S 10.20%.
Description and references
Antibiotic produced by the mold Aspergillus flavus from a Czapek-Dox medium supplemented with corn steep liquor or by Penicillium chrysogenum Q176 or by P. notatum strains: McKee, MacPhillamy, Proc. Soc. Exp. Biol. Med. 53, 247 (1943); Bush, Goth, J. Pharmacol. Exp. Ther. 78, 164 (1943); McKee et al., J. Bacteriol. 47, 187 (1944); Bush et al., J. Pharmacol. Exp. Ther. 84, 264 (1945); Fried et al., J. Biol. Chem. 163, 341 (1946); see also Wintersteiner under “Flavacidin” in Chemistry of Penicillin (Princeton, 1949). Prepn by hydrogenation of 2-pentenylpenicillin: Catch et al., GB 584852; Cook, Heilbron in Chemistry of Penicillin (Princeton, 1949). Characterization and antibacterial activity: Leigh, Nature 163, 95 (1949).
Derivative
Sodium salt.Nomenclature
CAS number: 575-47-3
Sodium n-amylpenicillinate; flavacidin; flavicin. C14H21N2NaO4S; mol wt 336.38.
C 49.99%, H 6.29%, N 8.33%, Na 6.83%, O 19.03%, S 9.53%.