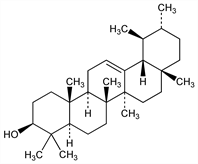
617. α-Amyrin
Nomenclature
Description and references
Occurs mostly as acetate in latex of rubber trees, in latex from Ficus variegata Blume, Moraceae, also in Balanophora elongata Blume, Balanophoraceae, and in Erythroxylum coca Lam. var. novogranatense Morris, and var. spruceanum Burck, Erythroxylaceae. Isoln from Manila elemi: Vesterberg, Westerlind, Ann. 428, 247 (1922). Structural studies: Spring, Vickerstaff, J. Chem. Soc. 1937, 249; Beynon et al., ibid. 1938, 1233; Meisels et al., Helv. Chim. Acta 32, 1075 (1949), 38, 1298 (1955); Melera et al., ibid. 39, 441 (1956). Identity with viminalol: Soldin, Marais, J. Pharm. Soc. 55, 452 (1966). Formation from ursolic acid: Goodson, J. Chem. Soc. 1938, 999; from boswellic acid: Ruzicka, Wirz, Helv. Chim. Acta 22, 948 (1939). Partial synthesis from glycyrrhetic acid and stereochemistry: Corey, Cantrall, J. Am. Chem. Soc. 81, 1745 (1959). Review: J. Simonsen, W. C. J. Ross, The Terpenes vol. IV (University Press, Cambridge, 1957) pp 116-148.