637. Androstenediol
Nomenclature
CAS number: 521-17-5
(3β,17β)-Androst-5-ene-3,17-diol; Δ5-androstene-3β,17β-diol. C19H30O2; mol wt 290.44.
C 78.57%, H 10.41%, O 11.02%.
Description and references
Obtained from dehydroandrosterone: Butenandt, Hanisch, Ber. 68, 1859 (1935); Z. Physiol. Chem. 237, 89 (1935); Ruzicka, Wettstein, Helv. Chim. Acta 18, 1264 (1935); FIAT Final Report 996, 45 (1947); Levy, Kapp, US 2521586 (1950 to Nopco Chem.).
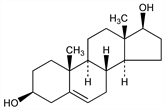
Properties
Leaflets from acetone + petr ether, or from methanol or ethyl acetate. Sublimes in high vacuum. mp 184°. [α]D18 55.5° (c = 0.4 in isopropanol). Insol in water.Derivative
3-Acetate.Nomenclature
CAS number: 1639-43-6
C21H32O3; mol wt 332.48.
C 75.86%, H 9.70%, O 14.44%.
Properties
Crystals from hexane, mp 147-148°.Derivative
17-Acetate.Nomenclature
CAS number: 5937-72-4
C21H32O3; mol wt 332.48.
C 75.86%, H 9.70%, O 14.44%.
Properties
Crystals from hexane, mp 146.5-148.5°, [α]D18 62.4° (alc).Derivative
Diacetate.Nomenclature
CAS number: 2099-26-5
C23H34O4; mol wt 374.51.
C 73.76%, H 9.15%, O 17.09%.
Properties
Leaflets from hexane, mp 165-166°, [α]D18 56.5° (alc).Derivative
17-Benzoate.C26H34O3; mol wt 394.55.
C 79.15%, H 8.69%, O 12.17%.
Properties
Crystals from methanol, mp 220-222°.Derivative
3-Acetate-17-benzoate.Nomenclature
CAS number: 5953-63-9
C28H36O4; mol wt 436.58.
C 77.03%, H 8.31%, O 14.66%.
Properties
Crystals, mp 180-182°.Derivative
Dipropionate.Nomenclature
CAS number: 2297-30-5
C25H38O4; mol wt 402.57.
C 74.59%, H 9.51%, O 15.90%.