636. Androstane-3β,11β-diol-17-one
Nomenclature
CAS number: 514-17-0
(3β,5α,11β)-3,11-Dihydroxyandrostan-17-one. C19H30O3; mol wt 306.44.
C 74.47%, H 9.87%, O 15.66%.
Description and references
First obtained by degradation of allopregnane-3β,11β,17α,20β,21-pentol (Reichstein's Substance A): Reichstein, Helv. Chim. Acta 19, 402 (1936), but later isolated in small quantities directly from extracts of the adrenal cortex: Reichstein, von Euw, ibid. 21, 1197 (1938); ibid. 24, 879 (1941). It is uncertain whether this substance occurs in the fresh adrenal gland; it may possibly occur by oxidation or decompn during the isolation procedure: Reichstein, Shoppee, Vitam. Horm. I, 368 (1943). Structure-activity study: S. Sassa et al., J. Biol. Chem. 254, 10011 (1979). Chromatographic studies: A. Kerebel et al., J. Chromatogr. 140, 229 (1977); J. T. Lin, E. Heftmann, ibid. 237, 215 (1982).
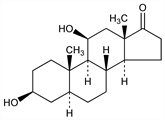
Properties
Needles from acetone + ether, mp 235-238°. [α]D20 +84.5° (ethanol); [α]D19 +81.3° (dioxane); [α]19545 +105° (dioxane). Precipitated by digitonin.Derivative
3-Acetate.C21H32O4; mol wt 348.48.
C 72.38%, H 9.26%, O 18.36%.
Properties
Needles from actone + ether, mp 230-231°. [α]D19 +70.5° (dioxane); [α]19546 +87.1° (dioxane).Derivative
Diacetate.C23H34O5; mol wt 390.51.
C 70.74%, H 8.78%, O 20.49%.