640. Androsterone
Nomenclature
CAS number: 53-41-8
(3α,5α)-3-Hydroxyandrostan-17-one; cis-androsterone; 3α-hydroxy-17-androstanone; androstan-3α-ol-17-one; 3α-hydroxyetioallocholan-17-one; 3-epihydroxyetioallocholan-17-one. C19H30O2; mol wt 290.44.
C 78.57%, H 10.41%, O 11.02%.
Description and references
Isolation from male urine after removal of the phenolic estrogen fraction: Butenandt, Tscherning, Z. Physiol. Chem. 229, 167 (1934); v. Euw, Reichstein, Helv. Chim. Acta 25, 988 (1942). Prepn from cholesterol: Ruzicka, ibid. 17, 1389 (1934); Marker, J. Am. Chem. Soc. 57, 1755 (1935); Schoeller et al., US 2232735 (1941 to Schering).
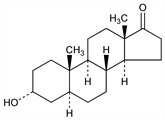
Properties
Crystals from acetone-ether, mp 185-185.5°. Sublimes in high vacuum. [α]D20 +94.6° (c = 0.7 in abs alc). [α]D15 +87.8° (c = 1.5 in dioxane). Not precipitated by digitonin. Barely soluble in water. Sol in most organic solvents.Derivative
Acetate.C21H32O3; mol wt 332.48.
C 75.86%, H 9.70%, O 14.44%.
Properties
Crystals from ether, sublimes in high vac, mp 165°, [α]D14 +76.7° (c = 2.04 in dioxane); [α]D25 +86° (c = 2 in ethanol).Derivative
Propionate.C22H34O3; mol wt 346.50.
C 76.26%, H 9.89%, O 13.85%.
Properties
mp 151-152°.Derivative
Benzoate.C26H34O3; mol wt 394.55.
C 79.15%, H 8.69%, O 12.17%.