644. Anethole Trithione
Nomenclature
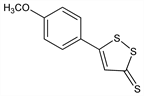
CAS number: 532-11-6
5-(4-Methoxyphenyl)-3H-1,2-dithiole-3-thione; 3-(p-anisyl)trithione; 3-(p-methoxyphenyl)-4,5-dithiacyclopent-2-ene-1-thione; trithio-p-methoxyphenylpropene; 5-(p-methoxyphenyl)-1,2-dithiacyclopent-4-ene-3-thione; 3-(p-methoxyphenyl)trithione; trithioanethole; anethole dithiolthione; ADT; Felviten (Grünenthal); Mucinol (Sanofi Winthrop); Sialor (Solvay); Sulfarlem (Solvay). C10H8OS3; mol wt 240.36.
C 49.97%, H 3.35%, O 6.66%, S 40.02%.
Description and references
Organosulfur compound that enhances glutathione synthesis and stimulates secretion of bile and saliva. Prepn: B. Bttcher, A. Lüttringhaus, Ann. 557, 89 (1947); O. Gaudin, N. Lozac'h, Compt. Rend. 224, 557 (1947); A. Thuillier, J. Vialle, Bull. Soc. Chim. Fr. 1959, 1398. HPLC determn in urine: A. N. Masoud, E. Bueding, J. Chromatogr. 276, 111 (1983). Clinical trial in xerostomia: J. B. Epstein et al., Oral Surg. Oral Med. Oral Pathol. 56, 495 (1983). Clinical trial in chemoprevention of lung cancer: S. Lam et al., J. Natl. Cancer Inst. 94, 1001 (2002). Review of mechanism of chemoprotective and antioxidant effects: M.-O. Christen, Methods Enzymol. 252, 316-323 (1995).
Properties
Orange-colored prisms from butyl acetate. Very bitter taste. mp 111°. Practically insol in water. Sol in pyridine, chloroform, benzene, dioxane, carbon disulfide. Slightly sol in ether, acetone, ethyl acetate, acetic acid, alc, cyclohexane, petr ether.Therapeutic Category
Choleretic; sialogogue.
Keywords
Choleretic; Sialagogue