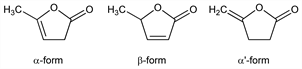
647. Angelica Lactone
Nomenclature
CAS number: 1333-38-6
5-Methyl-2-furanone. C5H6O2; mol wt 98.10.
C 61.22%, H 6.16%, O 32.62%.
Description and references
Exists in three forms. Prepn of α and β-forms: Wolff, Ann. 229, 250 (1885); Thiele, Ann. 319, 184 (1901); v. Auwers, Ber. 56, 1672 (1923); J. H. Helberger et al., Ann. 561, 215 (1949). Prepn of α′-form: J. P. Wineburg et al., J. Heterocycl. Chem. 12, 749 (1975); V. Jger, H. J. Günther, Tetrahedron Lett. 1977, 2543; R.A. Amos, J. A. Katzenellenbogen, J. Org. Chem. 43, 560 (1978). Toxicity data for α-form: E. J. Moran et al., Drug Chem. Toxicol. 3, 249 (1980).