646. Angelic Acid
Nomenclature
CAS number: 565-63-9
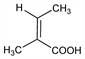
(2Z)-2-Methyl-2-butenoic acid; cis-2-dimethylcrotonic acid; 2-methylisocrotonic acid; cis-2,3-dimethylacrylic acid. C5H8O2; mol wt 100.12.
C 59.98%, H 8.05%, O 31.96%.
Description and references
Stereoisomer of tiglic acid. Found in ester form in sumbul root, Angelica archangelica L., Umbelliferae and together with tiglic acid esters in the oil of the Roman camomile, Anthemis nobilis L., Compositae. Isoln from seeds of Schoenocaulon officinale (Lindl.) A. Gray, Liliaceae (cevadilla seeds) by alkaline hydrolysis of cevadine: Stoll, Seebeck, Helv. Chim. Acta 35, 1275 (1952). Synthesis by trans addition of bromine to tiglic acid: Buckles, Mock, J. Org. Chem. 15, 680 (1950). Review and bibliography: Buckles et al., Chem. Rev. 55, 659 (1955).
Properties
Monoclinic rods, needles, plates; mp 45°. Spicy odor. Vesicant. d447 0.983. bp760 185°; bp12 86°. Sublimes. Volatile with steam. nD47 1.4434. pK (25°) 4.30. uv max (H2O): 217 nm (ε 5.15 × 103). Molar heat of combustion 626.6 kcal. Sparingly soluble in cold water, freely sol in hot water. Sol in alcohol, ether. Prolonged boiling of aq soln causes isomerization to tiglic acid; the process is speeded up by traces of bromine and sunlight, also by strong mineral acids or alks. Dry crystals of angelic acid have been stored in bottles for years without evidence of isomerization.Derivative
Calcium salt dihydrate.Ca(C5H7O2)2.2H2O; mol wt 274.32.
Ca 14.61%, C 43.78%, H 6.61%, O 34.99%.
Properties
Leaflets. Much more soluble in water than calcium tiglate: 100 parts of aq soln satd at 17.5° contains 23 parts of anhydr calcium angelate.Derivative
Amide.C5H9NO; mol wt 99.13.
C 60.58%, H 9.15%, N 14.13%, O 16.14%.
Properties
Crystals, mp 127-128°.Derivative
Methyl ester.C6H10O2; mol wt 114.14.
C 63.14%, H 8.83%, O 28.03%.
Properties
Liquid; d420 0.9413; bp764 128°; nD20 1.4321.Derivative
Ethyl ester.C7H12O2; mol wt 128.17.
C 65.60%, H 9.44%, O 24.97%.