658. Anileridine
Nomenclature
CAS number: 144-14-9
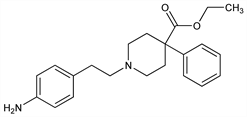
1-[2-(4-Aminophenyl)ethyl]-4-phenyl-4-piperidinecarboxylic
acid ethyl ester; 1-(p-aminophenethyl)-4-phenylisonipecotic acid ethyl
ester; ethyl 1-(4-aminophenethyl)-4-phenylisonipecotate; N-[β-(p-aminophenyl)ethyl]-4-phenyl-4-carbethoxypiperidine; N-β-(p-aminophenyl)ethylnormeperidine; Leritine (Merck & Co.). C22H28N2O2; mol wt 352.47.
C 74.97%, H 8.01%, N 7.95%, O 9.08%.
Description and references
Opioid analgesic. Synthesis: Weijlard et al., J. Am. Chem. Soc. 78, 2342 (1956); US 2966490 (1960 to Merck & Co.).
Properties
White crystalline powder. Oxidizes on exposure to air and light, becoming darker in color. mp 83°. Freely sol in alcohol, chloroform; sol in ether, although it may show turbidity. Very slightly sol in water.Derivative
Dihydrochloride.Nomenclature
CAS number: 126-12-5
C22H28N2O2.2HCl; mol wt 425.39.
C 62.12%, H 7.11%, N 6.59%, O 7.52%, Cl 16.67%.
Properties
Crystals from methanol + ether, mp 280-287° (dec). uv max (pH 7 in 90% methanol contg phosphate buffer): 235, 289 nm (A1%1cm 293, 34.5). Distribution coefficient (water, pH 3.6/n-butanol): 0.9. Freely soluble in water, methanol. Solubility in ethanol: 8 mg/g. pH of aq solns 2.0 to 2.5. Solns are stable at pH 3.5 and below. At pH 4 and higher the insol free base is precipitated.Note
This is a controlled substance (opiate): 21 CFR, 1308.12.Therapeutic Category
Analgesic.
Keywords
Analgesic; Opioids; Phenylpiperidines