Nomenclature
CAS number: 62-53-3
Benzenamine; aniline oil; phenylamine; aminobenzene; aminophen; kyanol.
C
6H
7N; mol wt 93.13.
C 77.38%, H 7.58%, N 15.04%.
Description and references
First obtained in 1826 by Unverdorben from
dry distillation of indigo. Runge found it in coal tar in 1834.
Fritzsche, in 1841, prepared it from indigo and potash and gave it
the name aniline. Manuf from nitrobenzene or chlorobenzene: Faith, Keyes & Clark's Industrial Chemicals, F. A. Lowenheim, M. K. Moran, Eds. (Wiley-Interscience, New York,
4th ed., 1975) pp 109-116. Procedures: A. I. Vogel, Practical Organic Chemistry (Longmans, London,
3rd ed., 1959) p 564; Gattermann-Wieland, Praxis des Organischen Chemikers (de Gruyter, Berlin,
40th ed., 1961) p 148. Brochure “Aniline” by Allied Chemical's
National Aniline Division (New York, 1964) 109 pp, gives reactions
and uses of aniline (877 references). Toxicity study: K. H. Jacobson, Toxicol. Appl. Pharmacol. 22,
153 (1972).
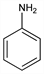
Properties
Oily liquid; colorless when freshly distilled,
darkens on exposure to air and light. Poisonous. Characteristic odor and burning taste; combustible; volatile with
steam. d2020 1.022. bp 184-186°. Solidif 6°. Flash pt, closed cup: 169°F (76°C). nD20 1.5863. pKb 9.30. pH of 0.2 molar aq soln 8.1. One gram dissolves in 28.6 ml water,
15.7 ml boil. water; misc with alcohol, benzene, chloroform, and most
other organic solvents. Combines with acids to form salts. It dissolves
alkali or alkaline earth metals with evolution of hydrogen and formation
of anilides, e.g., C6H5NHNa. Keep well closed and protected from light. Incompat. Oxidizers, albumin, solns of Fe, Zn, Al,
acids, and alkalies. LD50 orally in rats: 0.44 g/kg (Jacobson).Derivative
Hydrobromide.
Nomenclature
CAS number: 542-11-0
C
6H
7N.HBr; mol wt 174.04.
C 41.41%, H 4.63%, N 8.05%, Br 45.91%.
Properties
White to slightly reddish, crystalline powder, mp 286°. Darkens in
air and light. Sol in water, alc. Protect
from light.Derivative
Hydrochloride.
Nomenclature
CAS number: 142-04-1
C
6H
7N.HCl; mol wt 129.59.
C 55.61%, H 6.22%, N 10.81%, Cl 27.36%.
Properties
Crystals, mp 198°. d 1.222. Darkens in air and light. Sol in about 1 part water; freely
sol in alc. Protect from light.Derivative
Hydrofluoride.
Nomenclature
CAS number: 542-13-2
C
6H
7N.HF; mol wt 113.13.
C 63.70%, H 7.13%, N 12.38%, F 16.79%.
Properties
Crystalline powder. Turns gray on standing.
Freely sol in water; slightly sol in cold, freely in hot alc.Derivative
Nitrate.
Nomenclature
CAS number: 542-15-4
C
6H
7N.HNO
3; mol wt 156.14.
C 46.15%, H 5.16%, N 17.94%, O 30.74%.
Properties
Crystals, dec about 190°. d 1.36. Discolors in air and light. Sol in
water, alc. Protect from light.Derivative
Hemisulfate.
Nomenclature
CAS number: 542-16-5
C
6H
7N.H
2SO
4; mol wt 142.17.
C 50.69%, H 5.67%, N 9.85%, S 11.28%, O 22.51%.
Properties
Crystalline powder. d 1.38. Darkens on exposure to air and light.
One gram dissolves in about 15 ml water; slightly sol in alc. Practically
insol in ether. Protect from light.Derivative
Acetate.
Nomenclature
CAS number: 542-14-3
C
6H
7N.C
2H
4O
2; mol wt 153.18.
C 62.73%, H 7.24%, N 9.14%, O 20.89%.
Properties
Prepd from aniline and acetic acid: Vignon, Evieux, Bull. Soc. Chim. Fr. [4] 3, 1012 (1908). Colorless liquid. d 1.070-1.072. Darkens with age; gradually
converted to acetanilide on standing. Misc with water, alc.Derivative
Oxalate.
Nomenclature
CAS number: 591-43-5
(C
6H
7N)
2.C
2H
2O
4; mol wt 276.29.
C 60.86%, H 5.84%, N 10.14%, O 23.16%.
Properties
Prepd from aniline and oxalic acid in alc soln:
Hofmann, Ann. 47,
37 (1843). Triclinic rods from water, mp 174-175°. Readily sol in water; sparingly
sol in abs alc. Practically insol in ether.Caution
Poisoning may occur from inhalation,
skin penetration or ingestion. Potential symptoms of overexposure
are navy blue to black lips, tongue, mucous membranes; slate gray
skin; headache, nausea, vomiting, confusion, ataxia, vertigo, tinnitus,
weakness, disorientation, lethargy, drowsiness; dyspnea on effort;
methemoglobinemia, cyanosis, coma; tachycardia, heart blocks, arryhthmia,
shock; painful micturition, hematuria, hemoglobinuria, methemoglobinuria,
oliguria, renal insufficiency; cirrhosis. Direct contact may cause
eye irritation. Potential occupational carcinogen. See NIOSH Pocket Guide to Chemical Hazards (DHHS/NIOSH 97-140, 1997) p 18; Clinical
Toxicology of Commercial Products, R. E. Gosselin et al., Eds. (Williams & Wilkins, Baltimore, 5th ed., 1984)
Section III, pp 31-36.Use
Manuf dyes, medicinals, resins, varnishes, perfumes,
shoe blacks; vulcanizing rubber; as solvent. Hydrochloride used in
manuf of intermediates, aniline black and other dyes, in dyeing fabrics
or wood black.
