670. Anisomycin
Nomenclature
Description and references
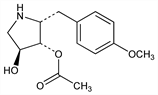
Protein synthesis inhibiting antibiotic isolated from Streptomyces: B. A. Sobin, F. W. Tanner, Jr., J. Am. Chem. Soc. 76, 4053 (1954); Tanner et al., US 2691618 (1954 to Pfizer). Activity: J. E. Lynch et al., Antibiot. Chemother. 4, 844, 899 (1954). Structure and stereochemistry: J. J. Beereboom et al., J. Org. Chem. 30, 2334 (1965); J. P. Schaefer, P. J. Wheatley, ibid. 33, 166 (1968); K. Butler, ibid. 2136. Biosynthesis: idem, ibid. 31, 317 (1966). Total synthesis: Oida, Ohki, Chem. Pharm. Bull. 16, 2086 (1968); ibid. 17, 1405 (1969); I. Felner, K. Schenker, Helv. Chim. Acta 53, 754 (1970). Chiral synthesis: J. P. H. Verheyden et al., Pure Appl. Chem. 50, 1363 (1978). Stereospecific total synthesis: D. P. Schumacher, S. S. Hall, J. Am. Chem. Soc. 104, 6076 (1982). Improved synthesis: A. N. Hulme, E. M. Rosser, Org. Lett. 4, 265 (2002). Mechanism of action: A. Jiménez, D. Vázquez in Antibiotics vol. 5(pt. 2), F. E. Hahn, Ed. (Springer-Verlag, New York, 1979) pp 1-19. Soly and stability data: Antibiot. Annu. 1954-55, pp 809-810. Memory impairment effects in mice: K. M. Lattal, T. Abel, Proc. Natl. Acad. Sci. USA 101, 4667 (2004).
Properties
Long needles from ethyl acetate or water, mp 140-141°. [α]D23 30° (methanol). uv max: 224, 277, 283 nm (ε 10800, 1800, 1600). pKa 7.9. Base is moderately sol in water; sol in lower alcohols, esters, ketones, chloroform; slightly sol in benzene, toluene and hexane.Derivative
Hydrochloride.Nomenclature
Properties
Crystals from ethyl acetate + ethanol, mp 187-188°. Very sol in water.Derivative
Deacetylanisomycin.Nomenclature
Description and references
Prepn from anisomycin: Nickell et al., US 2935444 (1960 to Pfizer).