679. Anserine
Nomenclature
CAS number: 584-85-0
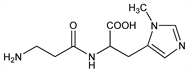
N-β-Alanyl-3-methyl-l-histidine; α-(β-aminopropionylamino)-β-(1-methyl-5-imidazolyl)propionic
acid; 3-methyl-N,α-(β-alanyl)-l-histidine. C10H16N4O3; mol wt 240.26.
C 49.99%, H 6.71%, N 23.32%, O 19.98%.
Description and references
First found in muscles of geese, later in many more animals, but not in man, cow, horse, or dog. Isoln: Ackermann, Z. Physiol. Chem. 183, 1 (1929); Wolff, Wilson, J. Biol. Chem. 109, 565 (1935). Synthesis: Behrens, du Vigneaud, J. Biol. Chem. 120, 517 (1937); Rinderknecht et al., J. Org. Chem. 29, 1968 (1964). Review: M. Guggenheim, Die biogenen Amine (Karger, Basel & New York, 4th ed., 1951) pp 439-446, 502.
Derivative
l-Form.Properties
Needles from dil alc, dec 240-242°. [α]D30 +12.3° (c = 5). pK1 2.64; pK2 7.04; pK3 9.49. The dried material is very hygroscopic. Freely soluble in water. Slightly sol in methanol, even less in ethanol.Derivative
Nitrate.C10H16N4O3.HNO3; mol wt 303.27.
C 39.60%, H 5.65%, N 23.09%, O 31.65%.