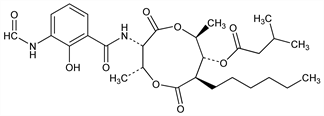
714. Antimycin A1
Nomenclature
CAS number: 642-15-9
3-Methylbutanoic acid 3-[[3-(formylamino)-2-hydroxybenzoyl]amino]-8-hexyl-2,6-dimethyl-4,9-dioxo-1,5-dioxonan-7-yl
ester; isovaleric acid 8-ester with 3-formamido-N-(7-hexyl-8-hydroxy-4,9-dimethyl-2,6-dioxo-1,5-dioxonan-3-yl)salicylamide. C28H40N2O9; mol wt 548.63.
C 61.30%, H 7.35%, N 5.11%, O 26.25%.
Description and references
Antibiotic substance produced by Streptomyces spp. Isoln: Dunshee et al., J. Am. Chem. Soc. 71, 2436 (1949); Lockwood et al., Phytopathology 44, 438 (1954); US 2657170 (1953 to Wisconsin Alumni Res. Found.). Structure: van Tamelen et al., J. Am. Chem. Soc. 83, 1639 (1961); Birch et al., J. Chem. Soc. 1961, 889. Absolute config.: Kinoshita et al., J. Antibiot. 25, 373 (1972).