751. Apraclonidine
Nomenclature
Description and references
α2-Adrenergic agonist; structural analog of clonidine, q.v. Synthesis: B. Rouot, G. LeClerc, Bull. Soc. Chim. Fr. Pt. 2, 520 (1979). Prepn (not claimed) and use in treatment of intraocular pressure: B. M. York, Jr., US 4517199 (1985 to Alcon). Pharmacology: B. Rouot et al., C. R. Seances Acad. Sci. Ser. D 286, 909 (1978). Receptor binding studies: D. C. U'Prichard, Prog. Clin. Biol. Res. 71, 53 (1981); D. C. Stump, D. E. MacFarlane, J. Lab. Clin. Med. 102, 779 (1983). Clinical pharmacology: D. A. Abrams et al., Arch. Ophthalmol. 105, 1205 (1987). Clinical evaluation in treatment of intraocular pressure: A. L. Robin et al., ibid. 1208; as additive to maximally tolerated medical therapy in glaucoma: A. L. Robin et al., Am. J. Ophthalmol. 120, 423 (1995).
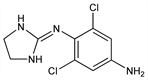
Properties
Solid, mp >230°.Derivative
Hydrochloride.Nomenclature
Properties
White powder. Sol in methanol; sparingly sol in water, alcohol. Insol in chloroform, ethyl acetate, hexanes.Derivative
Dihydrochloride.Nomenclature
Properties
uv max (ethanol): 254, 304 nm (ε 1800, 2500).Therapeutic Category
Treatment of post-surgical elevated intraocular pressure.
Keywords
α-Adrenergic Agonist; Antiglaucoma