786. Aristolochic Acid
Nomenclature
Description and references
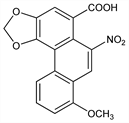
One of a group of fourteen known, substituted 1-phenanthrenecarboxylic acids, aristolochic acids, that occur in Aristolochiaceae and in butterflies feeding on these plants. They are often accompanied by aristololactams, twelve of which have been characterized. Isoln: Gnshirt, Pharmazie 8, 584 (1953); Pailer et al., Monatsh. Chem. 86, 676 (1955); Coutts et al., J. Pharm. Pharmacol. 11, 607 (1959); Kupchan, Doskotch, J. Med. Pharm. Chem. 5, 657 (1962). Structure: Pailer et al., Monatsh. Chem. 87, 249 (1956). Synthesis: Kupchan, Wormser, J. Org. Chem. 30, 3792 (1965). Biosynthesis: Spenser, Tiwari, Chem. Commun. 1966, 55. Acute toxicity: U. Mengs, Arch. Toxicol. 59, 328 (1987). Review: D. B. Mix et al., J. Nat. Prod. 45, 657-666 (1982).