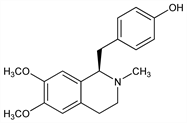
787. Armepavine
Nomenclature
CAS number: 524-20-9
4-[[(1R)-1,2,3,4-Tetrahydro-6,7-dimethoxy-2-methyl-1-isoquinolinyl]methyl]phenol; ()-6,7-dimethoxy-1-p-hydroxybenzyl-2-methyl-1,2,3,4-tetrahydroisoquinoline. C19H23NO3; mol wt 313.39.
C 72.82%, H 7.40%, N 4.47%, O 15.32%.
Description and references
Isoln from Papaver armeniacum (L.) DC., Papaveraceae: Konowalowa et al., Ber. 68, 2161 (1935); from Euonymus europaea L., Celastraceae: Bishay et al., J. Pharm. Pharmacol. 23 (Suppl), 233S (1971); from Rhamnus frangula L., Rhamnaceae: Pailer, Haslinger, Monatsh. Chem. 103, 1399 (1972). Structure: eidem, J. Gen. Chem. USSR 10, 641 (1940). Synthesis: Marion et al., J. Org. Chem. 15, 216 (1950); Gibson et al., J. Heterocycl. Chem. 3, 99 (1966). Resolution: Knabe, Horn, Arch. Pharm. 300, 547 (1967); Farber, Giacomazi, Chem. Ind. (London) 1968 (2), 57.
Properties
mp 149-150°. [α]D20 117.6° (CHCl3) (Knabe); [α]D27 103.2° (Farber).Derivative
10-Camphorsulfonate hydrate.C29H39NO7S.H2O; mol wt 563.70.
C 61.79%, H 7.33%, N 2.48%, O 22.71%, S 5.69%.
Properties
mp 130°. [α]D20 65° (MeOH).Derivative
dl-Form hydrate.Properties
Needles from acetone + ether; loses its water around 100°, then melts at 140°. Freely sol in alc, acetone, chloroform; slightly sol in ether. Almost insol in water.Derivative
dl-Form hydrochloride.C19H23NO3.HCl; mol wt 349.85.
C 65.23%, H 6.91%, N 4.00%, O 13.72%, Cl 10.13%.