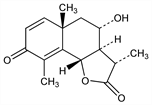
816. Artemisin
Nomenclature
CAS number: 481-05-0
(3S,3aR,4S,5aS,9bS)-3a,5,5a,9b-Tetrahydro-4-hydroxy-3,5a,9-trimethylnaphtho[1,2-b]furan-2,8(3H,4H)-dione; 6α,8α-dihydroxy-3-oxoeudesma-1,4-dien-12-oic acid 12,6-lactone; 8-hydroxysantonin. C15H18O4; mol wt 262.30.
C 68.68%, H 6.92%, O 24.40%.
Description and references
From the closed, unexpanded flower heads of several Artemisia spp, especially Artemisia maritima L., and A. cina Berg., Compositae (“wormseed”). Found in the mother liquors from the extraction of santonin. Isoln: E. Merck, E. Merck's Jahresber. 1894, 3; Chem. Zentralbl. 1895, I, 436. Structure: Sumi, J. Am. Chem. Soc. 80, 4869 (1958); Cocker, McMurry, Tetrahedron 8, 181 (1960). Stereochemistry: Bolt et al., J. Chem. Soc. 1963, 5235. Synthesis of (+)-artemisin: Nakazaki, Naemura, Tetrahedron Lett. 1966, 2615.