830. Ascorbic Acid
Nomenclature
Description and references
Physiological antioxidant. Coenzyme for a number of hydroxylation reactions; required for collagen synthesis. Widely distributed in plants and animals. Most primates (including humans), guinea pigs, and some birds and fish cannot synthesize ascorbic acid. Inadequate intake results in deficiency syndromes such as scurvy. Dietary sources include citrus fruits, potatoes, peppers, broccoli, cabbage, and rose hips. First isolated from the adrenal cortex of ox and later from lemons and paprika (originally called hexuronic acid): A. Szent-Gyrgyi, Biochem. J. 22, 1387 (1928); W. N. Haworth, A. Szent-Gyrgyi, Nature 131, 24 (1933). Structure studies: R. W. Herbert et al., J. Chem. Soc. 1933, 1270. Synthesis: R. G. Ault et al., ibid. 1419; T. Reichstein et al., Helv. Chim. Acta 16, 561, 1019 (1933); 17, 311, 510 (1934). Review of discovery, structure and synthesis: E. L. Hirst, Fortschr. Chem. Org. Naturst. 2, 132-159 (1939). Crystal structure: J. Hvoself, Acta Chem. Scand. 18, 841 (1964). Review of syntheses: T. C. Crawford, S. A. Crawford, Adv. Carbohydr. Chem. 37, 79-155 (1980). HPLC determn in plasma: J. Lykkesfeldt et al., Anal. Biochem. 229, 329 (1995). Discussion of use in the treatment of the common cold: L. Pauling, Proc. Natl. Acad. Sci. USA 68, 2678 (1971); H. Hemil, Scand. J. Infect. Dis. 26, 1 (1994). Clinical applications in immunology, lipid metabolism and cancer: Int. J. Vitam. Nutr. Res. 1982, Suppl. 23, 294 pp. Comprehensive description: I. A. Al-Meshal, M. M. A. Hassan, Anal. Profiles Drug Subs. 11, 45-78 (1982). Reviews: G. M. Jaffe in Kirk-Othmer Encyclopedia of Chemical Technology vol. 24 (John Wiley & Sons, New York, 3rd Ed., 1984) pp 8-40; “Vitamin C” in Vitamins, W. Friedrich, Ed. (Walter de Gruyter, Berlin, 1988) pp 929-1001. Review of pharmacology and clinical applications: M. Levine, N. Engl. J. Med. 314, 892-902 (1986); H. E. Sauberlich, Annu. Rev. Nutr. 14, 371-391 (1994).
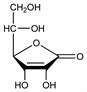
Properties
White crystals or powder (usually plates, sometimes needles, monoclinic system). Pleasant, sharp acidic taste. mp 190-192° (some dec). d 1.65. [α]D25 +20.5 to +21.5° (c = 1 in water); [α]D23 +48° (c = 1 in methanol). pH = 3 (5 mg/ml); pH = 2 (50 mg/ml); pK1 = 4.17; pK2 = 11.57. uv max at pH 2: 245 nm (E1%1cm 695); at pH 6.4: 265 nm (E1%1cm 940). Redox potential (first stage): E10 +0.166 V (pH 4). One gram dissolves in about 3 ml water, 30 ml alc, 50 ml abs alc, 100 ml glycerol, 20 ml propylene glycol. Soly in water: 80.0% at 100°; 40.0% at 45°. Insol in ether, chloroform, benzene, petr ether, oils, fats, fat solvents. Strong reducing agent. Stable to air when dry; aq solns are rapidly oxidized by air.Derivative
Calcium hypophosphite.Nomenclature
Asphocalcium; Calscorbat (Aerocid).Derivative
Calcium salt.Nomenclature
Description and references
Prepn: Ruskin, Merrill, Science 105, 504 (1947).
Properties
Dihydrate, triclinic crystals, [α]D20 +95.6° (c = 2.4). Freely sol in water. Practically insol in methanol, ethanol.Derivative
Sodium salt.Nomenclature
Description and references
Prepn: Holland, US 2442005 (1948).
Properties
Minute crystals, dec 218°. [α]D20 +104.4°. Soly in water: 62 g/100 ml at 25°; 78 g/100 ml at 75°.Use
As antimicrobial and antioxidant in foodstuffs.Therapeutic Category
Vitamin (antiscorbutic).
Therapeutic Category (Veterinary)
Vitamin (antiscorbutic).Keywords
Vitamin/Vitamin Source; Vitamin C