832. Asenapine
Nomenclature
Description and references
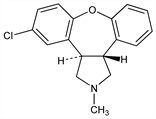
Combined serotonin (5HT2) and dopamine (D2) receptor antagonist; structurally related to mianserin, q.v. Prepn: BE 854915 (1977 to Akzo); W. J. van der Burg, US 4145434 (1979 to Akzona). Synthesis of labelled compd: J. Vader et al., J. Labelled Compd. Radiopharm. 34, 845 (1994). Series of articles on properties, metabolism and pharmacology: Arzneim.-Forsch. 40, 536-554 (1990). Receptor binding study: E. Richelson, T. Souder, Life Sci. 68, 29 (2000). Clinical pharmacology: B. Andrée et al., Psychopharmacology 131, 339 (1997). Review of development and therapeutic potential: J. J. Miguel-Hidalgo, Curr. Opin. Cent. Peripher. Nerv. Syst. Invest. Drugs 2, 85-92 (2000).
Properties
Log P at 37° (n-octanol/water): 6.33.Derivative
Maleate.Nomenclature
Properties
Racemic mixture. Crystals, mp 141-145°. uv max (ethanol): 270 nm (ε 1900). Soly at 21° (g/l): water 3, ethanol 30, methanol 250, acetone 125, ethyl acetate 10, dichloromethane 250, hexane <1. pH at 20° (0.1% soln in water): 4.56 ±0.05. pH at 23° (saturated soln 5.8 g/l): 4.43. pK1 <3, pK2 7.52, pK3 8.51.Therapeutic Category
Antipsychotic.
Keywords
Antipsychotic; Other Tricyclics; Serotonin-Dopamine Antagonist