837. Asparagine
Nomenclature
Description and references
Non-essential amino acid for human development. First amino acid noted in natural sources, it was named for the asparagus juice from which it was isolated: L. N. Vauquelin, P. J. Robiquet, Ann. Chim. 57, 88 (1806). Due to ease of conversion to aspartic acid, q.v., it was not isolated from protein until 1932: M. Damodoran, Biochem. J. 26, 235 (1932). Early chemistry and biochemistry: Amino Acids and Proteins, D. M. Greenberg, Ed. (Charles C. Thomas, Springfield, IL, 1951) 950 pp, passim; J. P. Greenstein, M. Winitz, Chemistry of the Amino Acids vols. 1-3 (John Wiley & Sons, Inc., New York, 1961) pp. 1856-1878, passim. As attachment point for saccharides: A. J. Mencke et al., Methods Enzymol. 138, 409 (1987). Colorimetric assay: S. Sheng et al., Anal. Biochem. 211, 242 (1993). Proposed formation of acrylamide, q.v., in cooking: D. S. Mottram et al., Nature 419, 448 (2002); R. H. Stadler et al., ibid. 449. Review of metabolism: D. A. Cooney, R. E. Handschumacher, Annu. Rev. Pharmacol. 10, 421-440 (1970); in plants: K. A. Sieciechowicz et al., Phytochemistry 27, 663-671 (1988). Review of deamidation in proteins: R. Bischoff, H. V. J. Kolbe, J. Chromatogr. B 662, 261-278 (1994).
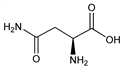
Properties
White, orthorhombic bisphenoidal crystals, mp 234-235° (bath preheated to 226°). d415 1.543. Acid to litmus. pK1 2.02; pK2 8.80. [α]D20 5.30° (c = 1.41); [α]D20 +34.26° (c = 2.24 in 3.4N HCl); [α]D20 6.35° (c = 11.23 in 2.5N NaOH). Sol in water, acids, alkalies. Practically insol in methanol, ethanol, ether, benzene.Derivative
d-Form.Nomenclature
Description and references
In peptidoglycans of bacterial cell walls: D. A. Reaveley, R. E. Burge, Adv. Microb. Physiol. 7, 1-81 (1972).