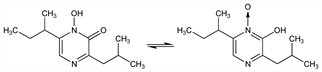
841. Aspergillic Acid
Nomenclature
Description and references
Antibiotic substance produced by Aspergillus flavus: White, Hill, J. Bacteriol. 45, 433 (1943); Dunn et al., J. Chem. Soc. 1949, Suppl. S126. Extraction and purification: Dutcher, J. Biol. Chem. 171, 321 (1947). Structure: idem, ibid. 232. 785 (1958). Biosynthesis: MacDonald, ibid. 236, 512 (1961), 237, 1977 (1962). Synthesis of racemate: Chigira et al., Bull. Chem. Soc. Jpn. 39, 632 (1966); Masaki et al., J. Org. Chem. 31, 4143 (1966); Ohta, Fujii, Chem. Pharm. Bull. 17, 851 (1969). Industrial prepn: Omata, Ueno, JP 65 13794 (1965 to Dainippon), C.A. 63, 11589b (1965). Proposed as a hypotensive: Jones, Martin, US 3720768 (1973 to Abbott). Reviews: MacDonald, “Aspergillic Acid and Related Compounds” in Antibiotics vol. II, D. Gottlieb, P. Shaw, Eds. (Springer-Verlag, New York, 1967) pp 43-51; Wilson, “Miscellaneous Aspergillus Toxins” in Microbial Toxins vol. VI, A. Ciegler et al., Eds. (Academic Press, New York, 1971) pp 207-235.