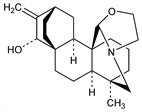
862. Atisine
Nomenclature
Description and references
From roots of the “atis” plant, Aconitum heterophyllum Wall., and from A. anthora L., Ranunculaceae: Broughton, Blue Book of East India Cinchona Cultivation 1877. 133; Goris, Metin, Compt. Rend. 180, 968 (1925); Lawson, Topps, J. Chem. Soc. 1937, 1640. Structure: Wiesner et al., Chem. Ind. (London) 1954, 132; Pelletier, Jacobs, J. Am. Chem. Soc. 76, 4496 (1954). Structure and stereochemistry: Dvornik, Edwards, Can. J. Chem. 42, 137 (1964). Partial synthesis: Pelletier, Jacobs, J. Am. Chem. Soc. 78, 4144 (1956); Pelletier, Parthasarathy, Tetrahedron Lett. 1963, 205. Racemic synthesis and resolution: Nagata et al., J. Am. Chem. Soc. 85, 2342 (1963); eidem, ibid. 89, 1499 (1967); Masamune, ibid. 86, 291 (1964); Guthrie et al., Tetrahedron Lett. 1966, 4645. 13C-NMR study of C-20 epimers: N. V. Mody, S. W. Pelletier, Tetrahedron 34, 2421 (1978).