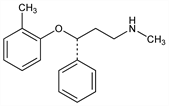
863. Atomoxetine
Nomenclature
Description and references
Norepinephrine reuptake inhibitor. Prepn (stereochem unspec): B. B. Molloy, K. K. Schmiegel, DE 2500110; eidem, US 4314081 (1975, 1982 both to Lilly). Prepn of (R)-form: B. J. Foster, E. R. Lavagnino, EP 52492 (1982 to Lilly); Y. Gao, K. B. Sharpless, J. Org. Chem. 53, 4081 (1988). Clinical pharmacokinetics: N. A. Farid et al., J. Clin. Pharmacol. 25, 296 (1985). Binding study: D. R. Gehlert et al., J. Neurochem. 64, 2792 (1995). Biotransformation by human liver microsomes: B. J. Ring et al., Drug Metab. Dispos. 30, 319 (2002). Evaluation of abuse potential: S. H. Heil et al., Drug Alcohol Depend. 67, 149 (2002). Clinical trial in pediatric ADHD: C. J. Kratochvil et al., J. Am. Acad. Child Adolesc. Psychiatry 41, 776 (2002); and comorbid tic disorders: A. J. Allen et al., Neurology 65, 1941 (2005). Review of clinical pharmacokinetics: J.-M. Sauer et al., Clin. Pharmacokinet. 44, 571-590 (2005).
Derivative
Hydrochloride.Nomenclature
Properties
Crystals, mp 166-168° (Foster, Lavagnino). [α]D25 38.01°; [α]36525 177.26° (c = 1 in methanol). Also reported as white solid from acetonitrile, mp 162-164° (Gao, Sharpless). [α]D23 41.37° (c = 1.02 in methanol); [α]D25 40.3° (c = 0.94 in ethanol). Soly in water: 27.8 mg/ml. pKa 10.13.Therapeutic Category
In treatment of attention deficit hyperactivity disorder (ADHD).