893. Azadirachtin
Nomenclature
Description and references
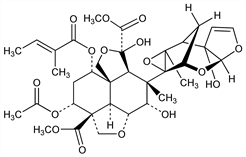
A tetranortriterpinoid isolated from the seeds of the neem tree, Azadirachta indica A. Juss. (Melia azadirachta L.), Meliaceae, and the chinaberry tree, M. azedarach L. Highly active insect feeding deterrent and growth regulator. Isoln from A. indica and identification as feeding inhibitor in locusts: J. H. Butterworth, E. D. Morgan, Chem. Commun. 1968, 23; from M. azedarach: E. D. Morgan, M. D. Thornton, Phytochemistry 12, 391 (1973). Total synthesis: G. E. Veitch et al., Angew. Chem. Int. Ed. 46, 7629 (2007). Structural studies: J. H. Butterworth et al., J. Chem. Soc. Perkin Trans. 2 1972, 2445. 1H- and 13C-NMR data and structure: P. R. Zanno et al., J. Am. Chem. Soc. 97, 1975 (1975); K. Nakanishi in Recent Advances in Phytochemistry vol. 9, V. C. Runeckles, Ed. (Plenum Press, New York, 1975) pp 283-298. Revised structure: W. Kraus et al., Tetrahedron Lett. 26, 6435 (1985); H. B. Broughton et al., Chem. Commun. 1986, 46. Isoln by HPLC: E. C. Uebel et al., J. Liq. Chromatogr. 2, 875 (1979); J. D. Warthen, Jr. et al., ibid. 7, 591 (1984). Antifeedant activity in locusts: J. S. Gill, C. T. Lewis, Nature 232, 402 (1971); in fall army worms, cotton bollworms: J. A. Klocke, I. Kubo, Entomol. Exp. Appl. 32, 299 (1982). Insect ecdysis and growth inhibition: H. Rembold, K. P. Sieber, Z. Naturforsch. 36C, 466 (1981); I. Kubo, J. A. Klocke, Agric. Biol. Chem. 46, 1951 (1982); K. P. Sieber, H. Rembold, J. Insect Physiol. 29, 523 (1983). Series of articles on chemistry and activity: Natural Pesticides from the Neem Tree, Proc. 1st Int. Neem Conf., 1980, H. Schmutterer et al., Eds. (German Agency for Technical Cooperation, Eschborn, 1981) 291 pp.