916. Azlocillin
Nomenclature
Description and references
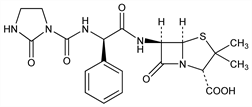
Semi-synthetic, broad-spectrum acylureido penicillin. Prepn: H. Disselnkotter, K. G. Metzger, FR 2100682; eidem, US 3933795 (1971, 1976 both to Bayer); H. B. Konig et al., Eur. J. Med. Chem. - Chim. Ther. 17, 59 (1982). In vitro studies: D. Stewart et al., Antimicrob. Agents Chemother. 11, 865 (1977). In vitro and in vivo activity: G. K. Daikos et al., Curr. Chemother., Proc. 10th Int. Congr. Chemother., 1977 (Amer. Soc. Microbiol., Washington, D.C., 1978) 1, pp 626-8. Pharmacokinetics: P. Fiegel, K. Becker, Antimicrob. Agents Chemother. 14, 288 (1978). Comparison with other penicillins: J. M. Andrews, K. A. Bedford, ibid. 559. Clinical studies: H. Lode et al., Infection 5, 163 (1977); E. B. Helm et al., Dtsch. Med. Wochenschr. 102, 1211 (1977). Series of articles on antibacterial activity, pharmacology, and clinical trials: Arzneim.-Forsch. 29, 1915-2032 (1979); Infection 10, Suppl. 3, S121-S266 (1982); J. Antimicrob. Chemother. 11, Suppl. B, 1-239 (1983).
Derivative
Sodium salt.Nomenclature
Properties
Pale yellow crystals, sol in water, methanol, DMF. Slightly sol in ethanol, isopropanol.Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Penicillins