937. Baclofen
Nomenclature
Description and references
Specific GABA-B receptor agonist. Prepn: NL 6407755; H. Keberle et al., US 3471548 (1965, 1969 both to Ciba). Toxicity study: T. Tadokoro et al., Osaka Daigaku Igaku Zasshi 28, 265 (1976), C.A. 88, 183016u (1978). Comprehensive description: S. Ahuja, Anal. Profiles Drug Subs. 14, 527-548 (1985). Review of pharmacology and therapeutic efficacy in spasticity: R. N. Brogden et al., Drugs 8, 1-14 (1974); of intrathecal use in spinal cord injury: K. S. Lewis, W. M. Mueller, Ann. Pharmacother. 27, 767-774 (1993). Clinical evaluation in reflex sympathetic dystrophy: B. J. van Hilten et al., N. Engl. J. Med. 343, 625 (2000). Benefit-risk assessment in severe spinal spasticity: A. Dario, G. Tomei, Drug Saf. 27, 799-818 (2004).
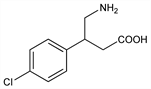
Properties
Crystals from water, mp 206-208°. Slightly sol in water; very slightly sol in methanol. Insol in chloroform. LD50 in male mice, rats (mg/kg): 45, 78 i.v.; 103, 115 s.c.; 200, 145 orally (Tadokoro).Derivative
Hydrochloride.Nomenclature
Properties
mp 179-181°.Therapeutic Category
Muscle relaxant (skeletal).
Therapeutic Category (Veterinary)
Muscle relaxant (skeletal).Keywords
Muscle Relaxant (Skeletal)