1078. Benzil
Nomenclature
CAS number: 134-81-6
Diphenylethanedione; bibenzoyl; dibenzoyl; diphenylglyoxal; diphenyl-α,β-diketone. C14H10O2; mol wt 210.23.
C 79.98%, H 4.79%, O 15.22%.
Description and references
Prepd by the oxidation of benzoin, C6H5CH(OH)COC6H5, with HNO3 or with a copper sulfate-pyridine mixture: Adams, Marvel, Org. Synth. vol. I, p 25 (1921); Clarke, Dreger, ibid. coll. vol. I, 80 (87, 2nd ed); Hatt et al., J. Chem. Soc. 1936, 93; L. F. Fieser, Experiments in Organic Chemistry (Boston, 3rd ed, 1955) p 173; Organic Experiments (Boston, 1964) p 214.
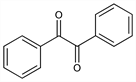
Properties
Yellow prisms (trigonal trapezohedral) from alcohol. d415 1.23. mp 95°. bp760 346-348°; bp12 188°. Absorption spectrum: Hantzsch, Schwiete, Ber. 49, 216 (1916). uv max (ethanol): 260 nm (ε 22000). Infrared in chloroform: 5.93; 6.22; 6.85 μ. Insol in water; sol in alcohol, ether, chloroform, ethyl acetate, benzene, toluene, nitrobenzene.Derivative
α-Benzilmonoxime.C14H11NO2; mol wt 225.24.
C 74.65%, H 4.92%, N 6.22%, O 14.21%.
Properties
Leaflets from 30% alc, mp 137-138°.Derivative
β-Benzilmonoxime.C14H11NO2; mol wt 225.24.
C 74.65%, H 4.92%, N 6.22%, O 14.21%.
Properties
Solvated needles from benzene, when dry mp 113-114°.Derivative
Disemicarbazone.C16H16N6O2; mol wt 324.34.
C 59.25%, H 4.97%, N 25.91%, O 9.87%.