1093. Benzoin
Nomenclature
CAS number: 119-53-9
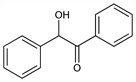
2-Hydroxy-1,2-diphenylethanone; benzoylphenylcarbinol; α-hydroxy-α-phenylacetophenone; bitter-almond-oil camphor. C14H12O2; mol wt 212.24.
C 79.23%, H 5.70%, O 15.08%.
Description and references
Prepd by treating an alcoholic soln of benzaldehyde with an alkali cyanide: Adams, Marvel, Org. Synth. vol. 1, p 33 (1921); coll. vol. I, 88; Arnold, Fuson, J. Am. Chem. Soc. 58, 1295 (1936); L. F. Fieser, Organic Experiments (D. C. Heath & Co., Boston, 1964) pp 211-214.
Derivative
dl-Form.Properties
Six-sided monoclinic prisms from alcohol, mp 137°. bp768 344°. bp12 194°. Reduces Fehling's soln. uv max (ethanol): 247 nm (ε 14500); infrared in chloroform: 2.88; 5.93; 6.21; 6.28, 6.85 μ. Soluble in 3335 parts water, more in hot water, in 5 parts pyridine; sol in acetone, in boiling alc; slightly in ether.Derivative
Methyl ether.C15H14O2; mol wt 226.27.
C 79.62%, H 6.24%, O 14.14%.
Properties
Needles, mp 49°.Derivative
Ethyl ether.C16H16O2; mol wt 240.30.
C 79.97%, H 6.71%, O 13.32%.