1116. Benzoyl Peroxide
Nomenclature
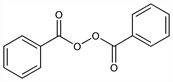
CAS number: 94-36-0
Dibenzoyl peroxide; benzoyl superoxide; Acetoxyl (Stiefel); Acnegel (Stiefel); Akneroxid L (Lubapharm); Benoxyl (Stiefel); Benzac (Galderma); Benzagel (RPR); Benzaknen (Galderma); Brevoxyl (Stiefel); Debroxide (Alcon); Desanden (Nycomed); Lucidol (Elf Atochem); Nericur (Schering); Oxy-5 (SKB); PanOxyl (Stiefel); Peroxyderm (Chassot); Persa-gel (J & J); Sanoxit (Galderma); Theraderm (BMS); Xerac BP (Person & Covey). C14H10O4; mol wt 242.23.
C 69.42%, H 4.16%, O 26.42%.
Description and references
Prepd by interaction of benzoyl chloride and a cooled soln of sodium peroxide. Laboratory procedure: A. I. Vogel, Practical Organic Chemistry (Longmans, London, 3rd ed., 1954) p 807; Gattermann-Wieland, Praxis des Organischen Chemikers (de Gruyter, Berlin, 40th ed., 1961) p 115. Comparative clinical study with clindamycin in acne vulgaris: L. J. Swinyer et al., Br. J. Dermatol. 119, 615 (1988). Review of carcinogenic and allergenic potential: D. J. Hogan, Int. J. Dermatol. 30, 467-470 (1991).
Properties
Crystals. mp 103-106°. May explode when heated. Sparingly sol in water or alcohol; sol in benzene, chloroform, ether. One gram dissolves in 40 ml carbon disulfide, in about 50 ml olive oil.Caution
Potential symptoms of overexposure are irritation of skin, eyes and mucous membranes; sensitization dermatitis. See NIOSH Pocket Guide to Chemical Hazards (DHHS/NIOSH 97-140, 1997) p 26.Use
Source of free radicals for industrial processes. Oxidizing agent in bleaching oils, flour, etc.; catalyst in the plastics industry; initiator in polymerization.Therapeutic Category
Keratolytic.
Therapeutic Category (Veterinary)
Keratolytic.Keywords
Antiacne; Keratolytic