Nomenclature
CAS number: 100-51-6
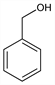
Benzenemethanol; phenylcarbinol; phenylmethanol; α-hydroxytoluene.
C
7H
8O; mol wt 108.14.
C 77.75%, H 7.46%, O 14.80%.
Description and references
Constituent of jasmine, hyacinth, ylang-ylang
oils, Peru and Tolu balsams, storax, where it occurs in ester form
also. Originally prepd by the Cannizzaro reaction from benzaldehyde
+ KOH: Cannizzaro, Ann. 88, 129 (1853); cf. Hickinbottom, Reactions of Organic Compds. (Longmans, London,
3rd ed., 1957) p 251; A. I. Vogel, Practical
Organic Chemistry (Longmans, London, 3rd ed., 1959) p
711; Gattermann-Wieland, Praxis des Organischen
Chemikers (de Gruyter, Berlin, 40th ed., 1961) p 193.
Produced on a large scale by the action of sodium or potassium carbonate
on benzyl chloride: DE 484662; Chem.
Zentralbl. 1930, I, 1052; Frdl. 16, 426; cf. Kirk-Othmer Encyclopedia of Chemical Technology vol. 3 (Interscience, New York, 1964) pp 442-449. Toxicity:
Smyth et al., Arch. Ind. Hyg.
Occup. Med. 4, 119 (1951).
Properties
Liquid. Faint aromatic odor. Sharp burning taste.
d420 1.04535; d425 1.04156. mp -15.19°. bp760 204.7°; bp400 183.0°; bp200 160.0°; bp100 141.7°; bp60 129.3°; bp40 119.8°; bp20 105.8°; bp10 92.6°; bp5 80.8°; bp1.0 58.0°. nD20 1.54035; nD25 1.53837:
Dreisbach, Martin, Ind. Eng. Chem. 41, 2875 (1941). Absorption spectrum: Brode, J. Phys. Chem. 30, 61 (1926).
Vapor density 3.72 (air = 1.00). Flash pt, closed cup 213°F, open cup 220°F. Autoignition temp 817°F. One gram dissolves
in about 25 ml water. One volume dissolves in 1.5 vols of 50% ethyl
alcohol. Misc with abs and 94% alcohol, ether, chloroform. LD50 orally in rats:
3.1 g/kg (Smyth).Use
Manuf other benzyl compds. Pharmaceutic aid (antimicrobial).
Organic solvent for gelatin, casein (when hot), solvent for cellulose
acetate, shellac. Used in perfumery and in flavoring (mostly in form
of its aliphatic esters). In microscopy as embedding material.
Therapeutic Category (Veterinary)
Has been used for relief from pruritis.
