Nomenclature
CAS number: 576-19-2
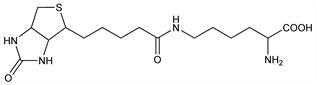
N6-[5-(Hexahydro-2-oxo-1
H-thieno[3,4-
d]imidazol-4-yl)-1-oxopentyl]-
l-lysine; ε-
N-biotinyl-
l-lysine; biotin complex of yeast.
C
16H
28N
4O
4S; mol wt 372.48.
C 51.59%, H 7.58%, N 15.04%, O 17.18%, S 8.61%.
Description and references
A naturally occurring complex of biotin. Contains
65.6% biotin. Isoln: Wright et al., J. Am. Chem. Soc. 72, 1048 (1950); 74, 1996 (1952). Structure: Peck et al., ibid. 72, 1048 (1950); 74, 1999 (1952). Synthesis: Wolf et al., ibid. 74, 2002 (1952); Weijlard et al., ibid. 76, 2505 (1954); Wolf, Folkers, US 2710298 (1955 to Merck & Co.). Purification:
McCormick, F"ory, Methods Enzymol. 18 (pt A), 413 (1970). Review: A. F. Wagner,
K. Folkers, Vitamins and Coenzymes (Wiley, New York, 1964) pp 138-159.
Properties
Crystals, mp 241-243°. Upon rapid crystn from dil methanol or dil
acetone, mp 228-230° (dec); upon slow crystn sinters at 227°. mp 245-252° (dec, microblock). Crystals from
water, mp 228.5°.
[α]D25 +53° (c = 1.05 in 0.1N NaOH). Infrared
absorption spectrum: J. Am. Chem. Soc. 74, 2001 (1952). Freely sol in water, glacial acetic
acid. Less sol in alc. Practically insol in acetone and most other
organic solvents. When subjected to strong acid hydrolysis (at least
3N at 120° for one hour) biocytin yields biotin and l-lysine. Forms a crystalline hydrochloride. Biocytin is characterized
microbiologically by its availability as a source of biotin to Lactobacillus casei, L. delbrückii LD 5, L.
acidophilus, Streptococcus fecalis R, Neurospora crassa, and Saccharomyces carlsbergensis and by its unavailability
as a source of biotin to Lactobacillus arabinosus, L. pentosus, and Leuconostoc mesenteroides P-60.