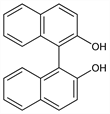
1226. BINOL
Nomenclature
Description and references
C2-symmetric ligand for metal-mediated stereoselective catalysis. Prepn: H. Walder, Ber. 15, 2166 (1882). Resolution of enantiomers: E. P. Kyba et al., J. Org. Chem. 42, 4173 (1977). Elucidation of catalyst structure: H. Sasai et al., J. Am. Chem. Soc. 115, 10372 (1993). Effect of substituents on enantioselectivity: C. Qian et al., J. Chem. Soc. Perkin Trans. 1 1998, 2097. Photochromism: M. Cavazza et al., J. Am. Chem. Soc. 118, 9990 (1996). Review of role in catalysis: R. Zimmer, J. Suhrbier, J. Prakt. Chem. 339, 758-762 (1997); with titanium: K. Mikami, Pure Appl. Chem. 68, 639-644 (1996); with rare-earth metals: H. Sasai et al., Chemtracts Org. Chem. 11, 264-267 (1998); of synthesis and use as a chiral reagent and ligand: J. M. Brunel, Chem. Rev. 105, 857-897 (2005).