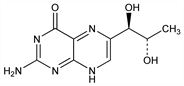
1229. Biopterin
Nomenclature
Description and references
A pteridine widely distributed in nature; naturally occurring as the l-erythro-form. Considered as a growth factor for some insects; see also Neopterin. Isoln from human urine: Patterson et al., J. Am. Chem. Soc. 77, 3167 (1955); 78, 5871 (1956); GB 814462 (1959 to Am. Cyanamid); from drosophila: H. S. Forrest, H. K. Mitchell, J. Am. Chem. Soc. 77, 4865 (1955); from queen bee jelly: Butenandt, Rembold, Z. Physiol. Chem. 311, 79 (1958). Absolute configuration: E. L. Patterson et al., J. Am. Chem. Soc. 78, 5871 (1956). Synthesis: E. L. Patterson et al., ibid. 5868; Tschesche et al., Ann. 658, 193 (1962); Rembold, Metzger, Ber. 96, 1395 (1963); Viscontini et al., Helv. Chim. Acta 55, 570, 574 (1972); B. Schircks et al., ibid. 60, 211 (1977); T. Sugimoto et al., Bull. Chem. Soc. Jpn. 53, 2344 (1980). Synthesis from neopterin: A. Kaiser, H. P. Wessel, Helv. Chim. Acta 70, 766 (1987). Biosynthesis: G. Kapatos et al., Science 213, 1129 (1981).