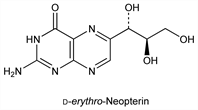
6462. Neopterin
Nomenclature
Description and references
Precursor in the biosynthesis of biopterin, q.v. Of the four possible isomers, d-erythro, l-erythro, d-threo, l-threo, two have been found in nature: the d-erythro form, to which the term neopterin originally referred; first isolated from the pupae of bees: Rembold, Buschmann, Ann. 662, 72 (1963); the l-threo form, found to be the growth factor for the protozoan Crithidia fasciculata and isolatable from cell-free extracts of Serratia indica: Kobashi, Iwai, Agric. Biol. Chem. 35, 47 (1971); 36, 1685, 1695 (1972). Both natural forms found in human urine: Fukushima, Shiota, J. Biol. Chem. 247, 4549 (1972). Early synthetic studies and structure: Rembold, Buschmann, Ber. 96, 1406 (1963). Synthesis of l-(-)-form: Viscontini, Provenzale, Helv. Chim. Acta 51, 1495 (1968). Synthesis of natural d-neopterin: Viscontini et al., ibid. 53, 1202 (1970).