1297. Bisphenol A
Nomenclature
CAS number: 80-05-7
4,4′-(1-Methylethylidene)bisphenol; 4,4′-isopropylidenediphenol; 2,2-bis(4-hydroxyphenyl)propane. C15H16O2; mol wt 228.29.
C 78.92%, H 7.06%, O 14.02%.
Description and references
Monomer used for polycarbonate and epoxy resins; exhibits estrogenic activity. Manuf from phenol and acetone: Jansen, US 2468982 (1949); Faith, Keyes & Clark's Industrial Chemicals, F. A. Lowenheim, M. K. Moran, Eds. (Wiley-Interscience, New York, 4th ed., 1975) pp 149-152. Identification as xenoestrogen released from polycarbonate: A. V. Krishnan et al., Endocrinology 132, 2279 (1993). HPLC determn in serum: K. Inoue et al., J. Chromatogr. B 749, 17 (2000); in food: H. Petersen et al., Eur. Food Res. Technol. 216, 355 (2003). Review of bioactivity and uses: N. Ben-Jonathan, R. Steinmetz, Trends Endocrinol. Metab. 9, 124-128 (1998); of properties and environmental fate: C. A. Staples et al., Chemosphere 36, 2149-2173 (1998).
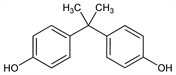
Properties
Crystals or flakes. Mild phenolic odor. mp 150-155° (solidification range). bp4 220°. Dec above 8 mm pressure when heated above 220°. Log P (n-octanol/water): 3.40. Soly in water: 120-300 mg/l. Sol in aq alkaline solns, alcohol, acetone. Slightly sol in carbon tetrachloride. LC50 (96 hr) in fathead minnow, rainbow trout: 4600, 3000-3500 μg/l (Staples).Derivative
Diglycidyl ether.Nomenclature
CAS number: 1675-54-3
BADGE. C21H24O4; mol wt 340.41.
C 74.09%, H 7.11%, O 18.80%.