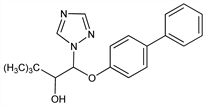
1305. Bitertanol
Nomenclature
CAS number: 55179-31-2
β-[(1,1′-Biphenyl)-4-yloxy]-α-(1,1-dimethylethyl)-1H-1,2,4-triazol-1-ethanol; 1-(biphenyl-4-yloxy)-3,3-dimethyl-1-(1H-1,2,4-triazol-1-yl)butan-2-ol; biloxazol; BAY KWG 0599; Baycor (Bayer); Sibutol. C20H23N3O2; mol wt 337.42.
C 71.19%, H 6.87%, N 12.45%, O 9.48%.
Description and references
Prepn: BE 814831; W. Kramer et al., US 3952002 (1974, 1976 both to Bayer AG). Physical properties and fungicidal activity: W. Brandes et al., Pflanzenschutz-Nachr. 32, 1 (1979). Mechanism of action: P. Kraus, ibid. 17; S. V. Overton et al., J. Hortic. Sci. 63, 183 (1988). GC determn in plants, soil and water: R. Brennecke, Pflanzenschutz-Nachr. 38, 33 (1985). Resolution of diastereomers: R. S. Burden et al., J. Chromatogr. 391, 273 (1987). In vitro activity: T. B. Sutton et al., Plant Dis. 69, 700 (1985). Field use in control of apple diseases: K. S. Yoder, ibid. 66, 580 (1982); W. F. S. Schwabe, Pflanzenschutz-Nachr. 35, 125 (1982); in wheat seed pretreatment: J. A. Hoffmann, D. V. Sisson, Plant Dis. 71, 839 (1987).