Description and references
Carotenoid carboxylic acids isolated from seeds
of Bixa orellana L., Bixaceae; principal pigments
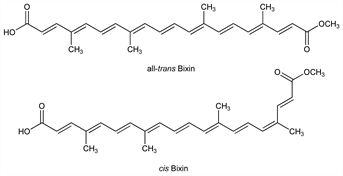
of the crude coloring extract, annatto, q.v. The natural product is the cis-form.
Isomerization to the more stable all-trans-form occurs
upon heating or commercial extraction. Isoln: J. B. Boussingault, Ann. Chim. Phys. 28, 440 (1825).
Isomerization studies: J. Herzig, F. Faltis, Ann. 431, 40 (1923); P. Karrer et al., Helv. Chim. Acta 12, 741 (1929). Structural studies: R. Kuhn et al., ibid. 11, 427 (1928); 12, 64, 904 (1929); Ber. 64, 1732 (1931); 65, 646, 1873 (1932). NMR determn of stereochemistry:
M. S. Barber et al., J. Chem.
Soc. 1961, 1625. Properties of bixin and norbixin:
J. F. Reith, J. W. Gielen, J. Food Sci. 36, 861 (1971). 1H and 13C NMR,
MS, and x-ray crystal structure determn: D. R. Kelly et al., J. Chem. Res. Miniprint 10, 2640 (1996). HPLC analysis of food coloring formulations:
M. J. Scotter et al., J. Agric.
Food Chem. 46, 1031 (1998). Biosynthesis and
prodn in genetically engineered E. coli (stereo unspecified):
F. Bouvier et al., Science 300, 2089 (2003). Review of cis-trans isomerization
and stereochemistry: L. Zechmeister, Chem.
Rev. 34, 267-344 (1944); of chemistry and extraction
from annatto: H. D. Preston, M. D. Rickard, Food Chem. 5, 47-56 (1980).
Derivative
cis-Form.
Nomenclature
CAS number: 6983-79-5
(2
E,4
Z,6
E,8
E,10
E,12
E,14
E,16
E,18
E)-4,8,13,17-Tetramethyl-2,4,6,8,10,12,14,16,18-eicosanonaenedioic
acid monomethyl ester; 9-
cis-6,6′-diapo-ψ,ψ-carotenedioic acid monomethyl
ester; 6′-methyl hydrogen (9′
Z)-6,6′-diapocarotene-6,6′-dioate; α-bixin; labile bixin.
C
25H
30O
4; mol wt 394.50.
C 76.11%, H 7.66%, O 16.22%.
Properties
Dark purple, lustrous crystals from ethanol +
chloroform or dichloromethane + acetone, mp 190 ± 1°; dec 200.3 ± 0.6°. Absorption max (chloroform): 502, 471 nm (log ε = 5.02, 5.07).Derivative
trans-Form.
Nomenclature
CAS number: 39937-23-0
(2
E,4
E,6
E,8
E,10
E,12
E,14
E,16
E,18
E)-4,8,13,17-Tetramethyl-2,4,6,8,10,12,14,16,18-eicosanonaenedioic
acid monomethyl ester; all
-trans bixin; β-bixin; isobixin; stable bixin.
C
25H
30O
4; mol wt 394.50.
C 76.11%, H 7.66%, O 16.22%.
Properties
Orange to purple plates from acetone, mp 204-206° (dec). Absorption max (chloroform): 507, 476 nm (E1%1cm 2970 ±40, 3240 ±50).
Sol in oil.Derivative
α-Norbixin.
Nomenclature
CAS number: 626-76-6
(2
E,4
E,6
E,8
E,10
E,12
E,14
E,16
Z,18
E)-4,8,13,17-Tetramethyl-2,4,6,8,10,12,14,16,18-eicosanonaenedioic
acid; 9-
cis-6,6′-diapo-ψ,ψ-carotenedioic acid.
C
24H
28O
4; mol wt 380.48.
C 75.76%, H 7.42%, O 16.82%.
Properties
No melting observed ≤280°; yellow coloring observed
at 240°; black coloring (carbonization) at ≥280°. Absorption max (chloroform): 499, 468 nm (E1%1cm 2200 ±30, 2470 ±30).Derivative
β-Norbixin.
Nomenclature
CAS number: 542-40-5
(All-
E)-4,8,13,17-tetramethyl-2,4,6,8,10,12,14,16,18-eicosanonaenedioic
acid; 6,6′-diapo-ψ,ψ-carotenedioic acid; all-trans norbixin.
C
24H
28O
4; mol wt 380.48.
C 75.76%, H 7.42%, O 16.82%.
Properties
No melting observed ≤300°; red color darkens ≥250°.
Absorption max (chloroform): 506, 475 nm; (0.1 N NaOH): 486, 457 nm. Sol in H2O.Food coloring; imparts a golden yellow color to
butter and cheese.