Nomenclature
CAS number: 507-70-0
(1
R,2
S,4
R)-
rel-1,7,7-Trimethylbicyclo[2.2.1]heptan-2-ol;
endo-1,7,7-trimethylbicyclo[2.2.1]heptan-2-ol;
endo-2-bornanol;
endo-2-camphanol;
endo-2-hydroxycamphane; (±)-borneol;
dl-borneol; bornyl alcohol; Baros camphor; Sumatra camphor; Borneo camphor; Dryobalanops camphor; Bhimsaim camphor; Malayan camphor; camphol.
C
10H
18O; mol wt 154.25.
C 77.87%, H 11.76%, O 10.37%.
Description and references
Constituent of essential oils of many plants.
Both d- and l- forms are naturally occurring.
The exo-isomer is known as isoborneol, q.v. Occurrence of d-form in oil from Dryobalanops aromatica Gaertn., Dipterocarpaceae, and l-form in Blumea balsamifera (L.) DC., Compositae: E. Gildemeister, F. Hoffman, Die therischen Ole (Schimmel, Leipzig, 3rd ed., 1928)
pp 475-481. Racemic borneol is prepd synthetically by reduction of
camphor: Truett, Moulton, J. Am. Chem. Soc. 73, 5913 (1951) DOI; Ziegler et al., Ann. 623, 9 (1959) DOI; Ziegler, GB 803178 (1958); from pinene: Schwyzer, Pharm. Ztg. 75, 1275 (1930). Configuration: Toivonen et al., Acta Chem. Scand. 3, 991 (1949) DOI. Review: J. L. Simonsen, The Terpenes vol. II (University
Press, Cambridge, 2nd ed., 1949) pp 349-365.
Derivative
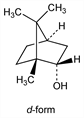
d-Form.
Nomenclature
CAS number: 464-43-7
(1
R,2
S,4
R)-borneol.
Properties
Hexagonal plates from petr ether, mp 208°. Peculiar peppery odor and
burning taste somewhat resembling that of mint. Sublimes, but is
less volatile than camphor. d
420 1.011. bp 212°. [α]
D20 +37.7° (c = 5 in alc); [α]
22546 +44.4° (c = 0.5 in toluene).
Flammable. Almost insol in water. Sol in alc (176 parts
dissolve in 100 parts w/w of abs alc), ether, petr ether (about 1:6),
benzene (about 1:5), toluene, acetone, decalin, tetralin. LD orally in rabbits: 2000 mg/kg,
see R. C. Beier,
Rev. Environ. Contam. Toxicol. 113, 47-127 (1990)
PubMed.
In flavors and fragrances; in the manuf of its esters.