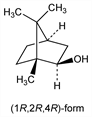
5128. Isoborneol
Nomenclature
Description and references
Prepn: Pickard, Littlebury, J. Chem. Soc. 91, 1973 (1907); Truett, Moulton, J. Am. Chem. Soc. 73, 5913 (1951) DOI; Ziegler, GB 803178 (1958). Resolution: Pickard, Littlebury, loc. cit.; Kenyon, Priston, ibid. 127, 1472 (1925). Prepn of the (1R,2R,4R)-form by reduction of d-camphor with lithium aluminum hydride: Trevoy, Brown, J. Am. Chem. Soc. 71, 1675 (1949) DOI. Configuration (isoborneol = exo-form; borneol = endo-form): Toivonen et al., Acta Chem. Scand. 3, 991 (1949) DOI. Separation of isoborneol from borneol via the p-nitrobenzoate deriv: Truett, Moulton, loc. cit. Review: J. L. Simonsen, The Terpenes vol. II (University Press, Cambridge, 2nd ed., 1949) pp 365-367; A. R. Pinder, The Chemistry of the Terpenes (Chapman & Hall, London, 1960) pp 22-24, 101, 103, 105-107, 111.