1414. 3-Bromo-d-camphor
Nomenclature
CAS number: 76-29-9
3-Bromo-1,7,7-trimethylbicyclo[2.2.1]heptan-2-one; 3-bromo-d-2-bornanone. C10H15BrO; mol wt 231.13.
C 51.97%, H 6.54%, Br 34.57%, O 6.92%.
Description and references
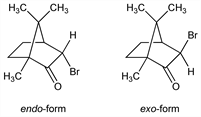
Of the two configurations found, the endo-form is more stable than the exo-form: Lowry et al., J. Chem. Soc. 121, 633 (1922); Cookson, ibid. 1954, 282. Prepn of the endo-form by bromination of d-camphor: Kipping, Pope, ibid. 63, 548 (1893); cf. Woods, Roberts, J. Org. Chem. 22, 1124 (1957). Prepn of exo-form by isomerization of endo-form: Lowry et al., loc. cit. Configuration: Wiebenga, Krom, Rec. Trav. Chim. 65, 663 (1946); Cookson, loc. cit. Review: J. L. Simonsen, Ed., The Terpenes vol. II (University Press, Cambridge, 2nd ed., 1949), pp 401-404.
Derivative
endo-Form.Nomenclature
α-Bromo-d-camphor; 3α-bromo-d-camphor; bromated camphor; camphor monobromated.Properties
Crystals from benzene, mp 76°. Camphor-like odor and taste. Discolors on prolonged exposure to light. d 1.449. [α]D20 +122.7° (14.5 g/100 g benzene soln), Cutter et al., J. Chem. Soc. 127, 1260 (1925). uv max (cyclohexane): 307.5 nm (log ε 1.98), Cookson, loc. cit. Sublimes, bp 274°. Almost insol in water: 1 g dissolves in 6.5 ml alcohol, 0.5 ml chloroform, 1.6 ml ether; sol in olive oil, slightly in glycerol. When phenol, chloral hydrate, salol, menthol, or thymol is triturated with bromocamphor the mixture melts; these compds, however, are not incompatible.Derivative
exo-Form.Nomenclature
α′-Bromo-d-camphor; 3β-bromo-d-camphor.Properties
Needles from methanol or ethanol, mp 78.5°. d 1.484. [α]D20 42.1° (14.5 g/100 g benzene soln), Cutter et al., loc. cit. uv max (cyclohexane): 312 nm (log ε 1.95), Cookson, loc. cit.Therapeutic Category
Topical counterirritant.